Medicoengineering cooperation and innovation translation are important pathways of progressing in clinical medicine. However, the translational road from an idea to a product is often long and rough, demanding not only arduous effort but also courage and persistence. Zhang Qiang from Nanfang Hospital, Southern Medical University, is such a young post-80s doctor having the above traits. In May 2021, the results of an animal study of through-the-scope twin clip (TTS-TC, nicknamed three-arm clip) for endoscopic closure of very large wounds invented by Zhang Qiang were published Gastrointestinal Endoscopy (GIE), an international authoritative journal in gastrointestinal endoscopy, attracting the attention and applause of peers.
However, it's rarely known that this invention has gone through ups and downs and there is an obscure story underlying it.
In the early 2019, the head of an upstream supplier called Leng Derong, the President of Micro-Tech, saying that one doctor named Zhang Qiang, referred by Department of Translational Medicine of Micro-Tech, asked to develop a product together. Manufacturing upstream parts only, this supplier is not familiar with medical device development, making it indeed very difficult to push the project forward after many setbacks. The head also said that, Zhang Qiang paid them more than RMB 300,000 as tooling charge, so if this project can proceed with the help of Micro-Tech, then the doctor's preliminary investment would not be lost in vain.
Although Micro-Tech has been involved in many medicoengineering cooperation projects since it was established, it is the first time to know such a case of investing so much money in prototype fabrication at the cost of a doctor. Immediately, Leng Derong found the director of Department of Translational Medicine and inquired about the case.
In fact, as early as in 2017, Zhang Qiang contacted the Department of Translational Medicine of Micro-Tech, requesting to translate a three-arm clip capable of full thickness suture. As arranged by Micro-Tech, Jin Hongyan, the No. 1 engineer who ever designed conceptual version of a hot product—SureClip, assisted Zhang Qiang in translating his proprietary conception into engineering drawings and fabricating the prototype. As the structure of TTS-TC is very complex, they worked for more than one year but failed to break through a few bottlenecks, and eventually, this project was not included in the development plan in that year.
Although Micro-Tech has high annual investment in R&D, which amounts to about RMB 100 million recently, budget regime is implemented in Micro-Tech, the project was not included in the budget and thus no expenditure was available. Nonetheless, Zhang Qiang was reluctant to give up the project and started toolmaking and trial fabrication at his own cost. Busy in clinical work, he was still mulling over how to improve the three-arm clip, and once any new idea emerged, he would call Jin Hongyan and the supplier's boss for long talks. Convinced by Zhang's dedication and persistence, Jin Hongyan continued to help Zhang improve the design and process until the end of 2018 when basic functions were achieved in the samples, and the next step should be application for registration certificate, which the supplier did not know how to do.
After learning this story, Leng Derong was much moved and immediately convened Zhang Bo, Marketing Vice President, and Li Changqing, R&D Vice President, to discuss together. All the participants were moved by the courage, persistence, and dedication of Zhang Qiang, and believed that this product would have excellent prospect. If the bottlenecks can be broken through using the state-of-the-art technology, then this product would be clinically very valuable—endosopy doctors would no longer be afraid of dealing with perforations, and secure support would be provided to exploration of Natural Orifice Transluminal Endoscopic Surgery (NOTES). So they decided unanimously to support Zhang Qiang and his TTS-TC project and it was agreed that, a down payment should be paid to Zhang Qiang so that he can get return from his earlier investment, moreover, he should obtain due short-term and long-term returns. A few days later, Zhang Bo flied to Guangzhou to meet Zhang Qiang and finalized the provisions of patent authorization together.

Later, Micro-Tech dispatched the No. 1 consumable development team, i.e., SureClip development team headed by Tang Zhi, to take over completely commercialization of TTS-TC and fully refine and optimize it ranging from design to process. Zhang Qiang also flied to Nanjing many times, where he conducted live animal testing at the Micro-Tech's Animal Testing Center to assess product performance and clinical effect. He and Tang's team gradually determined clinical needs and broke through the technical bottlenecks, and with efforts in more than one year, they eventually succeeded in trial fabrication of TTS-TC capable of opening and closing multiple times, 360-degree revolving, enabling single arm closing separately or both arms closing simultaneously and securely stretching to close giant wounds. Its design was closed in the first quarter of 2021 and evolved into procedures of application for China's registration certificate, CE in EU, and FDA 510K. It is expected to be marketed in the world in a few months to come.
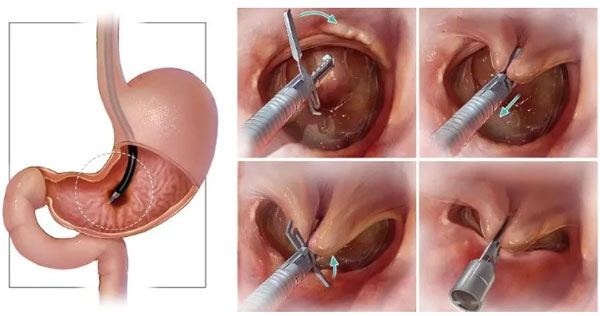
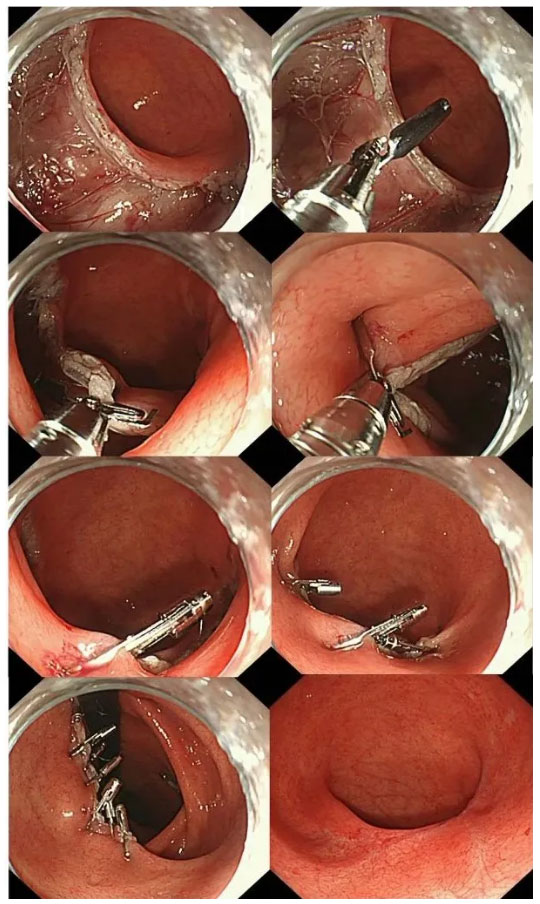
On May 7, 2021, Zhang Qiang's achievement in animal study of TTS-TC was published in GIE, an hilarious event!
In the past decade, considerable progresses have been made in China's medical device industry. But we must be clearly aware that, most of the progresses are built based on the achievements of innovations in developed countries. To put it boldly, simple and rough manufacturers imitate directly the innovations while slightly more motivated manufacturers make some improvements based on imitation. As Prof. Zhang Weiyin from Peking University said, this is a sort of arbitrage activity leveraging the achievements of previous innovations. But nowadays, everything imitable has been imitated, and once-huge arbitrage room has been almost used up. Therefore, China's medical device manufacturers have to move from the imitation and improvement-driven development to forefront innovation-driven development.
Innovations in medical devices have two modes in general: one is promotion by major scientific progresses, such as magnetic resonance and CT, and the other is promotion by medicoengineering cooperation, such as stents and flaps. Major scientific progresses are uncommon but medicoengineering cooperation can become normal.
To succeed in medicoengineering cooperation, the most crucial factor is to have doctors having innovative competence. Zhang Qiang, a post-80s, shows us the spirit of assiduous study, the competence of interdisciplinary research, the courage, and the confidence of new generation of doctors. In particular, his firm and indomitable will upon frustrations sheds light on success of medicoengineering cooperation in China.
As a partner in medicoengineering cooperation, Micro-Tech has innate gene of medicoengineering cooperation. From cupped ballhead stent for decreasing complication incidence to radiotherapeutic particle stent for extending the life of an advanced-stage patient, from the first single-use endoscopic biopsy forceps in China to the first indigo carmine dye in China, from SureClip disrupting the market status of old international brands to "golden" ESD knives close to clinical needs, from disruptive pancreatic cyst puncture system to cholangioscope setting up a new benchmark, and so on, such achievements of medicoengineering cooperation bring forth Micro-Tech today, and achievements of ongoing medicoengineering cooperation will surely bring forth Micro-Tech in future.
Let's look forward to seeing more doctors like Zhang Qiang!
 Call us on:
Call us on:  Email Us:
Email Us:  Micro-Tech (Nanjing) Co., Ltd
Micro-Tech (Nanjing) Co., Ltd